PPSU Tubing Medical Applications: Properties, Benefits, and Selection Guide
PPSU (polyphenylsulfone) tubing is a high-performance polymer used in medical devices that require repeated sterilization, chemical resistance, and long-term durability. It withstands over 1,000 steam autoclave cycles at 134°C, meets ISO 10993 and USP Class VI biocompatibility standards, and outperforms polycarbonate in hydrolytic stability. From surgical instruments to fluid management systems, PPSU tubing is a proven material for reusable and single-use healthcare products.
Medical device manufacturers face a common challenge: finding tubing materials that survive harsh sterilization cycles without losing strength or clarity. Traditional options like polycarbonate crack under repeated autoclaving, while metals add weight and cost. This is where PPSU tubing stands out. Polyphenylsulfone delivers the rare combination of high heat resistance, toughness, and chemical durability that modern medical applications demand. Whether you're designing reusable surgical tools, sterilization trays, or fluid delivery systems, PPSU tubing offers a reliable foundation. In this guide, we'll break down what makes PPSU tubing ideal for healthcare, how it compares to alternatives, and how to choose the right grade for your specific application.
What Is PPSU Tubing and Why Is It Used in Medical Devices?
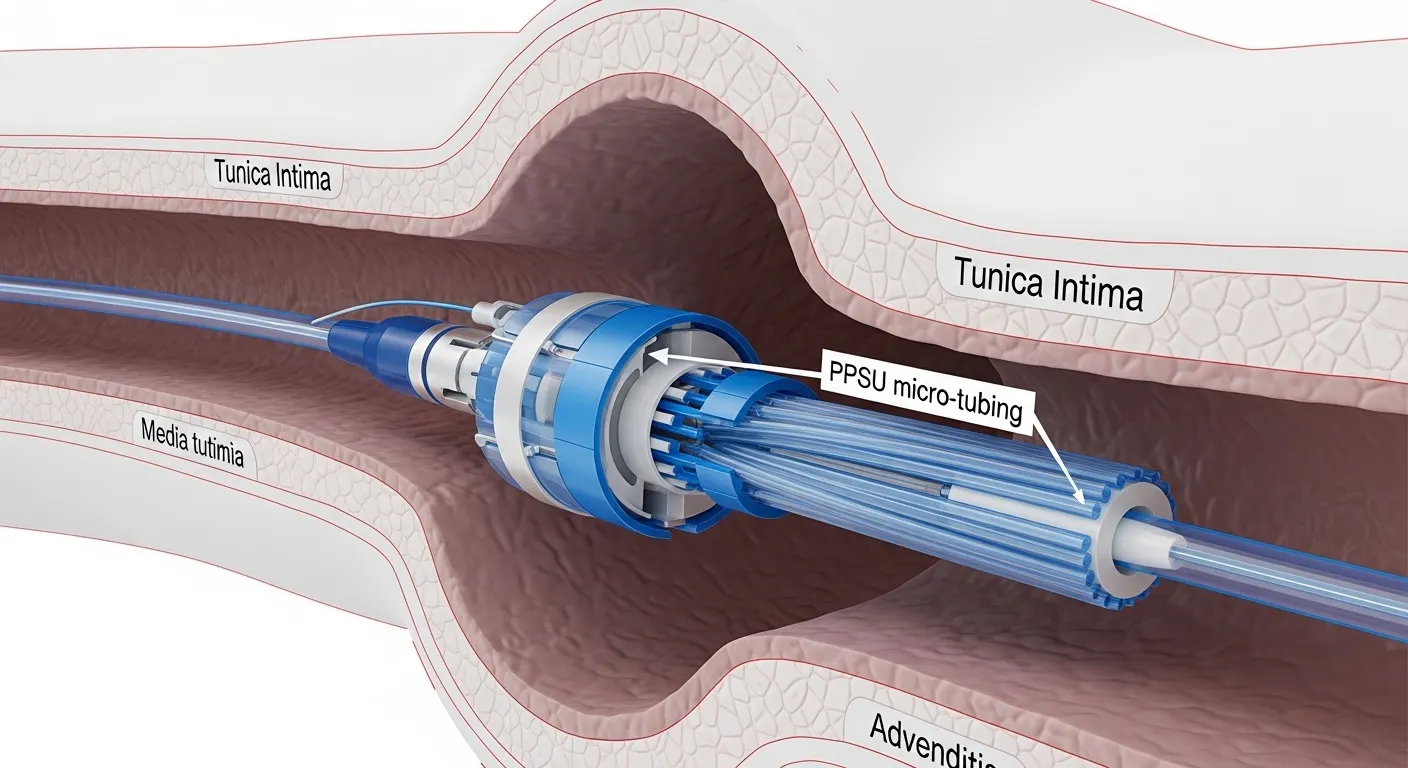
PPSU tubing is an extruded tube made from polyphenylsulfone, a high-performance amorphous thermoplastic known for its exceptional toughness, thermal stability, and resistance to hydrolysis. Medical device manufacturers use it for components that must survive repeated sterilization and chemical exposure without losing structural integrity.
What sets PPSU apart from standard medical-grade plastics is its glass transition temperature (Tg) of approximately 220°C (428°F). This means the material maintains its shape and mechanical properties at temperatures that would deform or degrade polycarbonate or standard polysulfone. Its heat deflection temperature (HDT) exceeds 204°C (400°F) under load, which directly translates to reliable performance inside autoclaves and high-temperature processing environments.
Manufacturers choose PPSU tubing for medical devices because it solves multiple problems at once. It resists the chemicals found in hospital disinfectants and sanitizers. It holds up to the mechanical stress of surgical handling. And it doesn't become brittle after hundreds of sterilization cycles, which gives medical devices a longer useful life and reduces replacement costs.
Key Properties That Make PPSU Tubing Ideal for Healthcare
PPSU tubing brings several material advantages that matter in clinical and manufacturing settings.
Thermal resistance is the standout property. With continuous operating temperatures around 204°C, PPSU tubing handles steam autoclaving at 134°C without warping, discoloration, or loss of mechanical integrity. This is critical for any tubing component that must be sterilized between uses.
Impact strength is another area where PPSU excels. The material has very high ductility, which means it absorbs shock without cracking. In surgical environments where instruments are handled quickly and sometimes dropped, this toughness prevents failures that could interrupt procedures.
Hydrolytic stability keeps the tubing strong over time. Unlike polycarbonate, which degrades when exposed to steam and moisture repeatedly, PPSU maintains its molecular structure through extended exposure to hot water and steam. This property directly extends the service life of reusable medical devices.
Chemical resistance protects against the aggressive cleaning agents used in hospitals. PPSU resists common disinfectants, sanitizers, and a range of organic solvents, which means it won't cloud, crack, or weaken during routine decontamination.
Dimensional stability ensures that tubing maintains consistent inner diameters and wall thicknesses across temperature changes. For applications like fluid delivery, where flow rates depend on precise tube geometry, this consistency is essential. Greetmed's approach to precision extrusion processes ensures that medical tubing meets these tight tolerances.
How Does PPSU Tubing Handle Repeated Sterilization?
PPSU tubing can withstand over 1,000 steam sterilization cycles at 134°C (273°F) while retaining its mechanical and visual properties. This makes it one of the most sterilization-resistant polymers available for medical tubing applications, far exceeding the limits of polycarbonate and standard polysulfone.
This performance matters because steam autoclaving remains the most common sterilization method in hospitals and surgical centers. Standard polysulfone (PSU) handles lower-temperature autoclaving at 121°C, but it struggles at the higher 134°C cycles that many facilities prefer for faster turnaround. PPSU bridges this gap comfortably.
Beyond steam, PPSU tubing is compatible with multiple sterilization methods used across the healthcare industry. These include ethylene oxide (EtO) gas sterilization, gamma radiation, and vaporized hydrogen peroxide (VHP). This versatility means device manufacturers can design products that work across different hospital sterilization protocols without worrying about material degradation.
The practical impact is significant. A PPSU tubing component that survives 1,000+ autoclave cycles reduces the total cost of ownership for hospitals. Devices last longer, replacement schedules stretch further, and procurement teams spend less on disposable alternatives. For manufacturers, it also means fewer warranty claims and stronger product reputation.
When selecting PPSU for sterilization-intensive applications, the key consideration is ensuring the specific grade has been tested and validated for the target sterilization method and cycle count. Not all PPSU grades perform identically, so material certificates and supplier testing data should be part of the evaluation process.
Where Is PPSU Tubing Used in Medical Devices?
PPSU tubing is used in surgical instrument components, sterilization trays, fluid handling systems, dental tools, catheter assemblies, and reusable device housings. Its combination of toughness, heat resistance, and biocompatibility makes it suitable for both patient-contact and non-contact applications across healthcare.
Here are the most common applications:
Surgical instrument components. PPSU's high impact strength and autoclave resistance make it a preferred material for instrument handles, grips, and housings. Surgical teams need tools that hold up to daily sterilization and the physical demands of the operating room. PPSU tubing and molded components deliver that durability without the weight of metal alternatives.
Sterilization trays and containers. Reusable sterilization trays made from PPSU maintain their structure after thousands of autoclave cycles. These trays organize and protect delicate instruments during the sterilization process, and PPSU ensures they don't warp or crack over time.
Fluid management systems. In devices that transport fluids, medications, or gases, PPSU tubing provides smooth internal surfaces and consistent flow paths. Its chemical resistance prevents degradation from contact with pharmaceutical solutions. This makes it a strong fit for connectors, couplings, and tubing segments in fluid delivery devices.
Catheter components. PPSU's high ductility positions it as a candidate for certain medical tube catheters and catheter sub-assemblies. While not used for the full catheter shaft in most designs, PPSU works well for connectors, hubs, and structural elements that require both flexibility and sterilization resistance.
Dental instruments. Dental tools face repeated autoclaving between patients, often multiple times per day. PPSU tubing and components in dental handpieces, suction lines, and instrument handles withstand this demanding cycle without degradation.
Reusable device housings. For devices designed to be reprocessed and reused, PPSU provides the structural integrity needed to survive years of clinical use. The material also allows manufacturers to design tri-layer medical tubing structures that combine PPSU with other polymers for optimized performance.
How Does PPSU Compare to Polycarbonate and PEEK for Medical Tubing?
PPSU outperforms polycarbonate in sterilization resistance and chemical durability, offers better impact strength than PEEK at a lower cost, and provides a practical middle ground for medical tubing applications that need high performance without the premium price of PEEK.
Here's how these three materials stack up:
PPSU vs. polycarbonate (PC). Polycarbonate has long been the go-to clear plastic for medical devices like blood oxygenators and drug delivery housings. However, PC cannot withstand steam sterilization and degrades under repeated autoclaving. PPSU tolerates 134°C steam cycles without issue. PC also lacks PPSU's resistance to hospital disinfectants and cleaning chemicals, which means PC components yellow, crack, or cloud over time in reprocessed devices. For any reusable medical tubing application, PPSU is the stronger choice.
PPSU vs. PEEK. PEEK offers higher rigidity and is often used in load-bearing structural applications like spinal implants. However, PEEK is opaque and significantly more expensive than PPSU. For non-structural tubing applications where toughness and sterilization resistance matter more than extreme rigidity, PPSU delivers comparable thermal performance at a fraction of the cost. PPSU also offers better impact resistance, which is important for tubing that needs to flex without fracturing.
PPSU vs. standard polysulfone (PSU). PSU and PPSU share a similar chemistry, but PPSU provides higher temperature resistance and significantly better impact strength. PSU handles autoclaving at 121°C, while PPSU tolerates 134°C. For applications requiring the higher sterilization temperature, PPSU is the clear upgrade.
The decision between these materials depends on your application requirements. Choose polycarbonate only for single-use, clear components that won't face steam sterilization. Choose PEEK for high-load structural implants. Choose PPSU for reusable tubing, instrument components, and fluid handling devices that need a balance of toughness, heat resistance, and cost efficiency.
Does Medical-Grade PPSU Meet Biocompatibility and Regulatory Standards?
Yes. Medical-grade PPSU resins are available in formulations that comply with ISO 10993 biocompatibility testing and USP Class VI requirements, confirming they are safe for medical devices with varying levels of patient contact. Certain PPSU grades also have FDA Master Access Files on record to simplify regulatory submissions.
ISO 10993 is the international standard that evaluates biological responses to medical device materials. It covers tests for cytotoxicity, sensitization, irritation, and systemic toxicity. Medical-grade PPSU formulations from major resin suppliers have completed these evaluations, giving device manufacturers a documented safety profile to reference in regulatory submissions.
USP Class VI testing takes this further by assessing material safety through implantation and injection studies. PPSU grades that pass USP Class VI are suitable for applications where the tubing may contact tissue, blood, or other body fluids during clinical use.
For manufacturers targeting the U.S. market, certain PPSU resin families (such as Radel PPSU from Syensqo) have FDA Master Access Files on record. This simplifies the regulatory pathway because device manufacturers can reference the existing material data rather than generating it from scratch.
The key consideration is sourcing medical-grade PPSU specifically formulated for healthcare use. Industrial-grade PPSU may have different additives or processing histories that affect biocompatibility. Always request material certificates, lot traceability, and compliance documentation from your tubing supplier.
How Do You Select the Right PPSU Tubing for Your Application?
Start by defining your sterilization method, chemical exposure profile, target dimensions, and regulatory pathway. Then match these requirements to a specific PPSU grade and work with an experienced medical tubing supplier to validate the material for your device's intended use and production volume.
Here's a practical selection framework:
Define your sterilization requirements. If the device will face steam autoclaving at 134°C, confirm that the PPSU grade is rated for your target cycle count. If EtO or gamma is the primary method, ensure the grade has been validated for those processes as well.
Specify your tube dimensions. Inner diameter, outer diameter, and wall thickness directly affect flow rates, flexibility, and burst pressure. Medical tubing demands tight tolerances, often within ±0.05 mm. Work with a manufacturer experienced in precision extrusion to ensure consistent geometry across production runs.
Evaluate chemical exposure. List every chemical the tubing will contact during its lifecycle: medications, cleaning agents, disinfectants, and body fluids. Cross-reference this list against the PPSU grade's chemical resistance data. While PPSU handles most common hospital chemicals well, certain aggressive solvents may require additional testing.
Confirm regulatory needs. Determine whether your device requires ISO 10993, USP Class VI, or both. Request documentation from the resin supplier and the tubing extruder. If you're pursuing FDA clearance, check whether the PPSU grade has an existing Master Access File.
Consider secondary operations. PPSU tubing can be machined, bonded, and assembled into complex devices. If your design requires flaring, welding, or overmolding, verify that the selected grade supports these processes without compromising material properties.
Partner with an experienced supplier. Medical tubing requires more than just material knowledge. Your supplier should understand extrusion tolerances, cleanroom requirements, and regulatory documentation. Greetmed offers a full range of medical consumables and custom tubing solutions designed for OEM medical device projects.
Conclusion
PPSU tubing gives medical device manufacturers a material that combines the heat resistance needed for demanding sterilization protocols, the toughness required for surgical environments, and the chemical durability to withstand years of hospital cleaning routines. It outperforms polycarbonate in reusable applications and offers a more cost-effective alternative to PEEK for non-structural components.
When evaluating PPSU tubing for your next project, focus on three things: match the grade to your sterilization method, confirm biocompatibility documentation for your regulatory pathway, and work with a tubing supplier that understands medical extrusion tolerances.
Ready to explore PPSU tubing for your medical device application? Contact Greetmed to discuss your custom tubing requirements with our engineering team.
Frequently Asked Questions
What temperature can PPSU tubing withstand during autoclaving?
PPSU tubing tolerates steam autoclaving at 134°C (273°F) and can endure over 1,000 sterilization cycles at this temperature without significant loss of mechanical properties. This makes it one of the most autoclave-resistant polymers available for medical tubing, far exceeding the 121°C limit of standard polysulfone.
Is PPSU tubing biocompatible for patient-contact medical devices?
Yes. Medical-grade PPSU formulations meet ISO 10993 and USP Class VI biocompatibility standards, confirming safety for devices that contact tissue, blood, or body fluids. Always verify that your specific PPSU grade carries the appropriate certifications for your device classification and intended use.
What is the difference between PPSU and polysulfone (PSU) tubing?
PPSU and PSU share similar chemistry, but PPSU offers higher temperature resistance and significantly better impact strength. PSU handles autoclaving at 121°C, while PPSU tolerates 134°C. PPSU also has superior hydrolytic stability, meaning it retains its properties over more sterilization cycles than PSU.
Can PPSU tubing be used in single-use medical devices?
Yes. While PPSU is often chosen for reusable devices because of its sterilization durability, it also works well in single-use applications that require chemical resistance, high clarity, or exposure to gamma radiation sterilization. The material's toughness and dimensional stability make it a strong option for single-use fluid management components and connectors.
How does PPSU tubing compare to polycarbonate in medical applications?
PPSU outperforms polycarbonate in steam sterilization resistance, chemical durability, and long-term hydrolytic stability. Polycarbonate cannot withstand repeated autoclaving and degrades when exposed to hospital disinfectants over time. For any reusable medical device that requires steam sterilization, PPSU is the recommended alternative.
Submit Your Request
Recent Posts
Tags
- Adult Diapers
- Are custom medical devices safe
- Baby Diapers
- Can respiratory anesthesia be used
- Digital Healthcare
- Do you offer customized consumables
- European Market
- How do you take care of a skin wound
- Industry Trends
- Lady Sanitary Napkins
- Medical Devices
- OEM Medical Devices
- Product Introductions
- Protective Equipment
- Under Pads
- What are custom-made medical devices
- What are diagnostic products
- What are hospital dressing products
- What are medical tube catheters
- What are some common protective equipment
- What are the appropriate applications for hospital dressing products
- What are the appropriate uses for protective equipment
- What is a gynecological examination
- What is a medical consumable
- What is an anesthesia kit
- What is an OEM in medical devices
- what is an wound skin care
- what is can disposable ultrasonic diagnostic
- What is good manufacturing medical devices
- What is hospital-grade protective equipment
- what is medical equipments hospital furniture
- What is medical sterilization wrapping
- What is rehabilitation equipment device
- What medical consumables do you supply
- Where can I find laboratory consumables wholesale
- where can I find medical protection device
- where to buy hypodermic accessories
- where to buy medical apparel
- where to buy medical consumable accessories
- where to find OEM medical device supplier
- where to find rehabilitation equipment supplier