Best Peel-Away Introducer Sheath Manufacturer | Expert Guide
Finding a reliable peel-away introducer sheath manufacturer is a critical step for medical distributors and healthcare facilities looking to ensure patient safety during vascular access. These devices are essential for placing catheters, pacing leads, and other medical components, as they allow for easy removal without displacing the inserted device. Choosing the right partner involves evaluating material quality, French size variety, and compliance with international medical standards.
What is a Peel-Away Introducer Sheath and Why is it Essential?
A peel-away introducer sheath is a specialized medical tube used to facilitate the percutaneous insertion of devices into the venous or arterial system. It features a unique "scored" body that allows the clinician to split the sheath symmetrically into two halves. This "peeling" action enables removal while the catheter or lead remains securely in place.
Manufacturers design these tools to minimize trauma during insertion. They typically consist of a high-quality PTFE or polyethylene tube, a dilator, and a guide wire. The ability to peel the sheath away is particularly vital in procedures like permanent pacemaker implantation or peripherally inserted central catheter (PICC) lines, where the hub of the device is too large to pass through a standard fixed sheath.
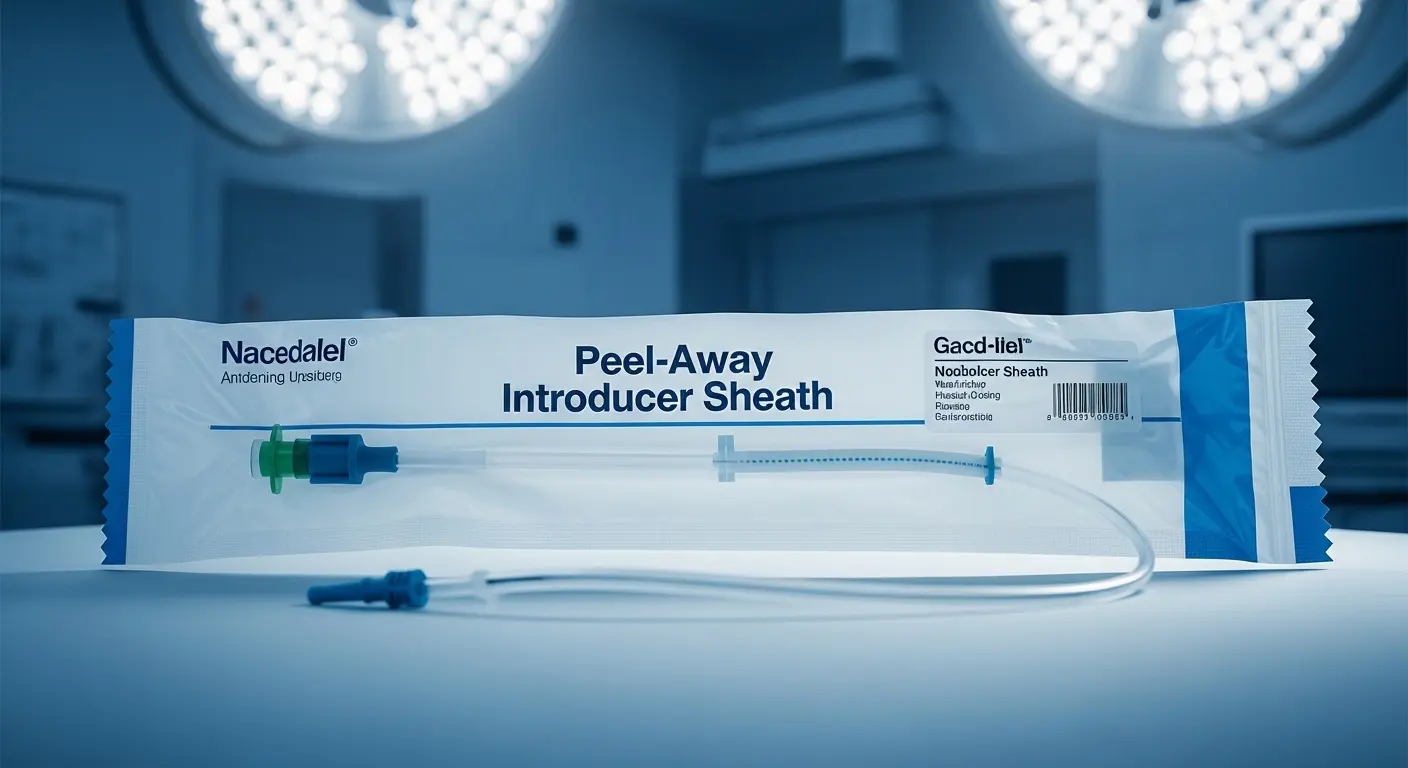
Key Components of a High-Quality Sheath System
| Component | Function | Material Importance |
| Sheath Tube | Provides the conduit for the device. | Must be kink-resistant and easy to split. |
| Vessel Dilator | Tapers the entry point for smooth access. | Requires a smooth transition to the sheath tip. |
| Guide Wire | Navigates the vessel path. | Needs specific flexibility and radiopacity. |
How Do You Choose the Best Peel-Away Introducer Sheath Manufacturer?
To choose the best peel-away introducer sheath manufacturer, prioritize companies with ISO 13485 certification and CE or FDA clearing. Look for a partner that offers a wide range of French sizes (typically 4F to 16F) and provides customizable lengths. A reputable manufacturer will also offer transparent data regarding their sheath’s "peel force" consistency and tip transition smoothness.
Expert buyers often look for manufacturers that control the entire production line. When a company handles the extrusion of the PTFE tubing and the molding of the hub in-house, they maintain tighter tolerances. This results in a sheath that doesn't "zip" prematurely but splits easily when the clinician applies intentional force.
Critical Selection Criteria
- Material Biocompatibility: Ensure the manufacturer uses medical-grade materials that reduce the risk of thrombogenicity.
- Hub Design: Ergonomic wings help the clinician grip and peel the device with one hand if necessary.
- Valve Options: Some manufacturers provide valved peel-away sheaths to prevent air embolism and blood loss during the procedure.
- Sterilization Standards: Verify if they use EO (Ethylene Oxide) sterilization and provide valid shelf-life data.
What Features Define a High-Performance Peel-Away Sheath?
A high-performance peel-away sheath features a low-profile design and a smooth transition between the dilator and the sheath tip to reduce insertion resistance. It must possess high kink resistance to maintain a clear lumen even in difficult anatomical turns. The "peel" must be symmetrical and require consistent force from the hub down to the distal tip.
I once worked with a clinical team that struggled with sheaths that would "jag" or tear unevenly. This can lead to jagged edges that might irritate the vessel wall during removal. Top-tier manufacturers solve this by precision-scoring the plastic. This ensures that the two halves separate like a zipper, providing a clean break every single time.
Technical Specifications to Look For
- French Size Accuracy: Internal and external diameters must be precise to match medical hypodermic syringe sizes or catheter gauges.
- Radiopacity: The tip or the entire sheath should be visible under fluoroscopy.
- Coating: Some advanced models feature hydrophilic coatings to make the initial insertion almost effortless.
Why is Symmetrical Peeling Critical in Clinical Settings?
Symmetrical peeling is critical because it prevents the sheath from "whipping" or tugging on the newly placed catheter as it is removed. If one side of the sheath is tougher than the other, the clinician may accidentally pull the lead out of position while trying to force the split. A balanced peel ensures the device remains stationary.
In the operating room, stability is everything. When a manufacturer gets the wall thickness exactly right, the clinician feels a steady resistance. This predictable feedback allows for a smooth exit. If you are ordering for a high-volume cardiac lab, testing a sample for "peel feel" is a smart move before committing to a large contract.
What Are the Most Common Sizes Provided by Manufacturers?
Manufacturers typically provide peel-away sheaths in sizes ranging from 4 French to 16 French, with lengths between 5cm and 25cm. The 6F, 7F, and 8F sizes are the most common for standard venous access, while larger sizes are reserved for dialysis catheters or bulky pacing leads. Custom lengths are often available for pediatric or specific deep-vein applications.
Common Size Chart for Peel-Away Sheaths
| French Size (F) | Inner Diameter (mm) | Typical Application |
| 4F - 5F | 1.35 - 1.67 | Pediatric PICC lines |
| 6F - 7F | 2.0 - 2.3 | Standard CVC and pacing leads |
| 8F - 10F | 2.6 - 3.3 | Large bore catheters |
| 12F+ | 4.0+ | Dialysis and advanced vascular tools |
When stocking your inventory, consider the variety of procedures your facility performs. For instance, knowing what hospital dressing products are used post-procedure can help you bundle the right sheath sizes with the necessary aftercare kits.
How Does Manufacturing Quality Impact Patient Safety?
Manufacturing quality directly impacts patient safety by reducing the risk of air embolisms, vessel trauma, and infection. A poorly manufactured sheath might have burrs at the tip or a dilator that doesn't fit snugly, causing a "step-off" that tears the vein during entry. High-quality manufacturers use cleanroom environments (Class 100,000 or better) to prevent contamination.
Using a sub-par peel-away introducer sheath manufacturer can lead to costly complications. If a sheath kinks during a procedure, the clinician might have to start over, increasing the time the patient is under stress. Reliable brands ensure their medical cotton swabs and access tools meet the same rigorous purity standards to keep the surgical field sterile.
What is the Difference Between Valved and Non-Valved Peel-Away Sheaths?
The main difference lies in the presence of a hemostatic valve that blocks the backflow of blood and prevents air from entering the venous system. Valved peel-away sheaths are preferred for patients at high risk of air embolism or in procedures where the sheath stays in the vessel for an extended period before the catheter is ready.
Many manufacturers now offer "hybrid" valves that can still be peeled away. While a standard malecot catheter might not require a complex valve for every use, vascular access often does. If a clinician specifies a "PRN" (as needed) requirement for valved options, it pays to have a manufacturer that stocks both. Understanding what PRN means in medical terms helps in managing these inventory requests effectively.
Frequently Asked Questions About Peel-Away Sheath Sourcing
Can I get custom-labeled sheaths for my brand?
Yes, many top manufacturers offer OEM (Original Equipment Manufacturer) services. They can print your logo on the hub and provide custom packaging that meets your specific regulatory needs.
What is the typical shelf life of a sterile peel-away sheath?
Most manufacturers guarantee a shelf life of 3 to 5 years, provided the packaging remains intact and stored in a cool, dry place away from direct sunlight.
Are these sheaths reusable?
No. Peel-away introducer sheaths are strictly single-use devices. The peeling mechanism destroys the integrity of the tube, and the materials are not designed to withstand resterilization processes like autoclaving.
How do I verify a manufacturer's credentials?
Ask for their ISO 13485 certificate and their latest audit report. You should also check if their products are registered in the specific countries where you plan to sell or use them.
Next Steps for Sourcing Your Medical Sheaths
When you are ready to contact a peel-away introducer sheath manufacturer, start by requesting a sample kit. Test the "peel force" yourself and check the smoothness of the dilator transition. A partner who is willing to provide detailed technical data and samples is usually one you can trust for a long-term supply.
Submit Your Request
Recent Posts
Tags
- Adult Diapers
- Are custom medical devices safe
- Baby Diapers
- Can respiratory anesthesia be used
- Digital Healthcare
- Do you offer customized consumables
- European Market
- How do you take care of a skin wound
- Industry Trends
- Lady Sanitary Napkins
- Medical Devices
- OEM Medical Devices
- Product Introductions
- Protective Equipment
- Under Pads
- What are custom-made medical devices
- What are diagnostic products
- What are hospital dressing products
- What are medical tube catheters
- What are some common protective equipment
- What are the appropriate applications for hospital dressing products
- What are the appropriate uses for protective equipment
- What is a gynecological examination
- What is a medical consumable
- What is an anesthesia kit
- What is an OEM in medical devices
- what is an wound skin care
- what is can disposable ultrasonic diagnostic
- What is good manufacturing medical devices
- What is hospital-grade protective equipment
- what is medical equipments hospital furniture
- What is medical sterilization wrapping
- What is rehabilitation equipment device
- What medical consumables do you supply
- Where can I find laboratory consumables wholesale
- where can I find medical protection device
- where to buy hypodermic accessories
- where to buy medical apparel
- where to buy medical consumable accessories
- where to find OEM medical device supplier
- where to find rehabilitation equipment supplier