Minimally Invasive Delivery System OEM: Precision Solutions
Why does a billion-dollar medical device project stall in the final stages of clinical trials? It’s rarely the "big idea" that fails. Instead, it's often the minimally invasive delivery system OEM partner that couldn't maintain tolerances or scaled too slowly.
In the high-pressure environment of minimally invasive surgery (MIS), the delivery system is the bridge between a surgeon’s intent and a patient’s recovery. If that bridge isn't perfectly engineered, the procedure fails. As we move deeper into 2026, the demand for delivery systems that are thinner, more flexible, and smarter is reaching a fever pitch. Here is why choosing the right OEM isn't just a procurement choice—it’s a strategic life-or-death decision.
The Evolution of the Delivery System OEM
A few years ago, an OEM was just a contract manufacturer. You gave them a drawing, and they gave you a part. Today, a minimally invasive delivery system OEM must be a co-designer. Modern MIS procedures—from neurovascular coiling to transcatheter aortic valve replacement (TAVR)—require systems with contradictory properties.
You need a proximal end that’s stiff enough to push through calcified arteries, but a distal tip that’s soft enough not to perforate a vessel. You need a shaft that’s thin enough to fit through a micro-incision but strong enough to house complex pull-wires and sensors. Achieving this balance requires a partner who understands the "physics of the catheter" as well as the "economics of the factory."
The Core Tech: Braiding, Coiling, and Multi-Lumen Extrusion
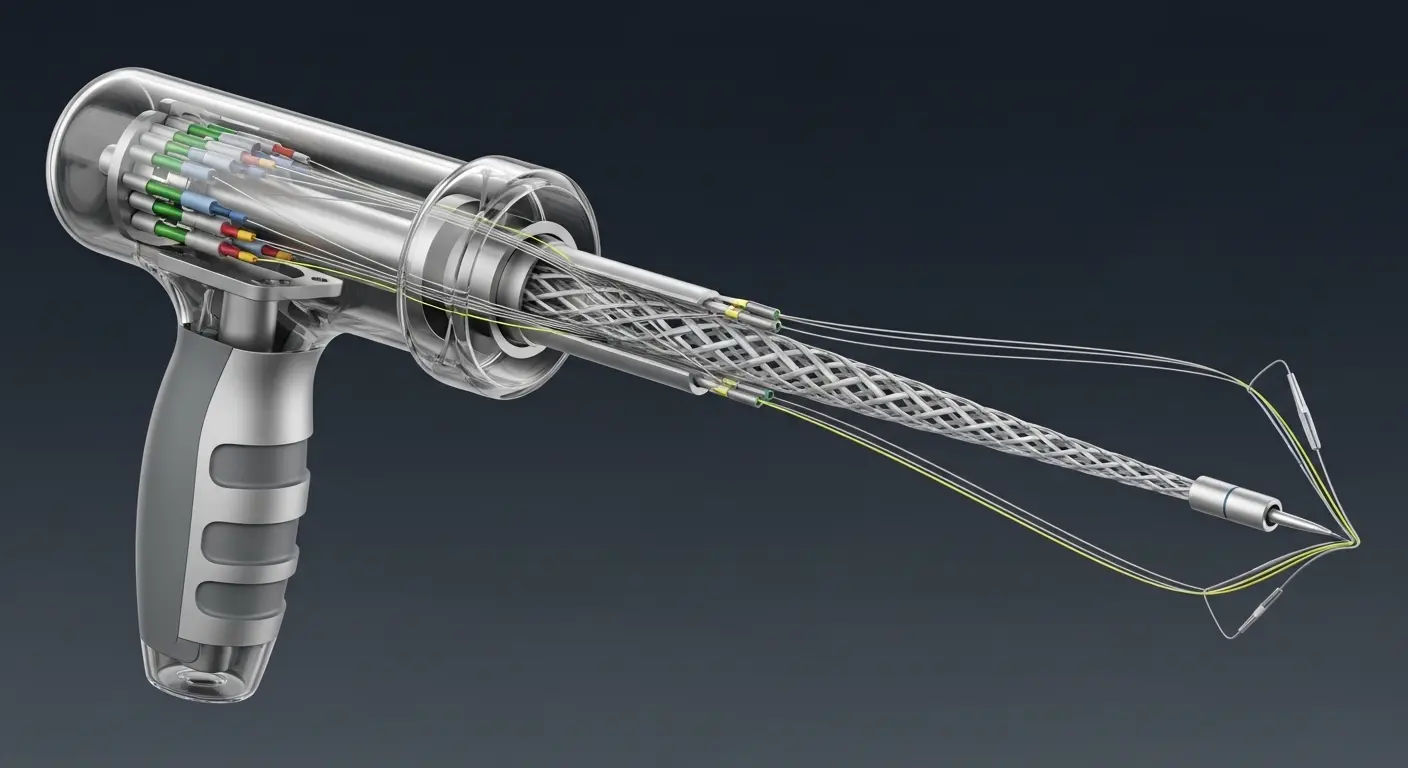
The "skeleton" of a delivery system determines its performance. At Greetmed, we focus on three technical pillars to ensure your device navigates the body with zero resistance.
1. Precision Multi-Lumen Extrusion Modern delivery systems often carry multiple tools at once. One lumen might carry a guidewire, while another carries a contrast agent, and a third houses a fiber-optic sensor. We utilize closed-loop extrusion lines to maintain tolerances as tight as 0.01mm. This ensures that every lumen is perfectly centered, preventing the "wall collapse" that can happen in inferior designs.
2. Variable Pitch Braiding "Pushability" is useless without "Torquability." If a surgeon rotates the handle 45°, the distal tip must rotate exactly 45°. We use variable pitch braiding (stainless steel or nitinol) to reinforce the tube. By changing the density of the braid along the shaft, we can create a device that transitions seamlessly from a rigid handle to a floppy, atraumatic tip.
3. Advanced Tip Forming and Bonding The most dangerous part of any catheter is the "Transition Zone." This is where the stiff braid ends and the soft tip begins. If this bond is weak, the tip can detach inside the patient. We use RF-tipping and laser-welding to create molecular-level bonds between dissimilar polymers like Pebax® and TPU.
This level of structural integrity is what separates a world-class delivery system from a basic surgical tube. Much like the precision required for manufacturing a high-quality medical hypodermic syringe, every micron of the delivery system must be accounted for.
Design for Manufacturability (DFM): Saving Time and Millions
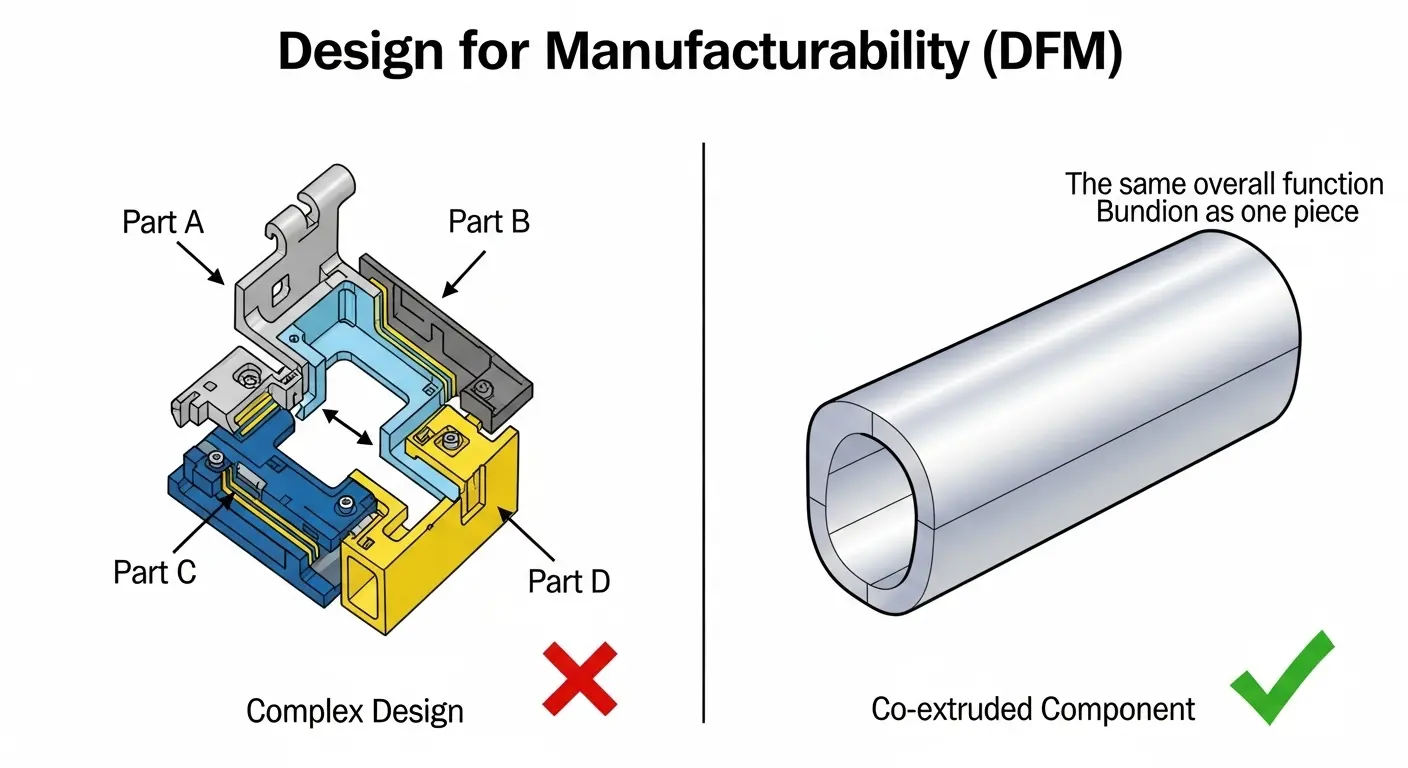
Most engineers design for the lab, not the line. A minimally invasive delivery system OEM should catch these errors before they cost you six months in rework.
When we review a design, we look for "The Complexity Tax."
- Part Consolidation: Can we replace three bonded components with one co-extruded tube?
- Material Selection: Are you using an exotic polymer that has a six-month lead time when a standard medical-grade Nylon 12 would perform better?
- Tolerance Optimization: Are you over-specifying a tolerance that adds 40% to the cost without improving clinical outcomes?
By applying these DFM principles early, we help our partners move from concept to prototype in weeks, not months. This efficiency is a core part of our philosophy, whether we are building a robotic delivery system or producing high-volume medical cotton swabs for global distribution.
The Role of Robotics and Smart Catheters in 2026

The biggest trend we’re seeing this year is the rise of robotic-assisted delivery systems. These systems require a different type of OEM expertise. In a robotic procedure, the "feel" of the catheter is replaced by haptic feedback sensors.
This means the delivery system must be incredibly consistent. A robot cannot "compensate" for a sticky catheter the way a human surgeon can. Our manufacturing processes include 100% in-line inspection using laser micrometers to ensure that the friction coefficient of every shaft is identical.
Furthermore, we are seeing a massive surge in "Smart" delivery systems—devices that include embedded chips or sensors to measure blood pressure or temperature in real-time. Integrating these electronics into a 2mm tube is the ultimate test for an OEM. It requires a cleanroom environment that can handle both traditional plastic extrusion and delicate electronic assembly.
Quality Systems: Beyond the ISO 13485 Sticker
Every OEM says they are "Quality First." But in 2026, regulators are demanding more than just a certificate. They want to see Process Capability (CpK) data.
When you partner with a minimally invasive delivery system OEM, you should ask: "Can you prove your process is stable?" At Greetmed, we operate under a "Total Quality" framework. We don't just inspect the final part; we monitor the "Environmental Variables" of the cleanroom. If the humidity in our Class 100,000 cleanroom fluctuations by 5%, our systems alert our engineers, as this can affect the curing of the medical-grade adhesives used in distal tip bonding.
This obsession with quality extends to our entire catalog. Whether you are sourcing a complex neurovascular delivery system or looking for hospital dressing products to stock a high-volume facility, the validation paperwork is just as rigorous.
FAQ: Navigating the OEM Partnership
What is the average lead time for a delivery system prototype? For a "Design-to-Print" project, we typically deliver prototypes in 4–6 weeks. If the project requires custom tooling or exotic materials, it may extend to 8–10 weeks.
How do you handle IP protection? As a dedicated OEM, your intellectual property is our most valuable asset. We use localized data silos and strict NDA protocols to ensure that your design innovations stay yours.
Can you handle Class III medical devices? Yes. Our facilities and quality systems are designed to meet the rigorous clinical data and validation requirements of high-risk Class III devices, including those used in cardiology and neurology.
Achieving this often requires specialized geometries, such as the malecot catheter, where structural stability and drainage performance must be balanced perfectly.
What does "PRN" mean when ordering components? In clinical logistics, some items are stocked as "as needed." If you’ve ever wondered what PRN means in medical terms, it essentially allows for flexible supply chain management in hospitals. As an OEM, we help our clients manage these "as needed" surges by maintaining a robust buffer of raw materials.
What is the difference between an OEM and a CM? While a CM (Contract Manufacturer) simply builds to your specs, an OEM (Original Equipment Manufacturer) like Greetmed often provides the engineering expertise to improve the design, handle the regulatory filing, and manage the sub-tier supply chain.
Why China-Greetmed is the Future of Global MedTech
The medical world is shifting. The need for high-quality devices is growing, but so is the pressure to reduce healthcare costs. This is the "Greetmed Advantage." We provide the technical precision of a high-end CDMO with the manufacturing efficiency of a global leader.
Precision doesn't have to be prohibitively expensive. By optimizing the minimally invasive delivery system OEM process, we help MedTech innovators bring their life-saving ideas to market faster and more affordably.
Submit Your Request
Recent Posts
Tags
- Adult Diapers
- Are custom medical devices safe
- Baby Diapers
- Can respiratory anesthesia be used
- Digital Healthcare
- Do you offer customized consumables
- European Market
- How do you take care of a skin wound
- Industry Trends
- Lady Sanitary Napkins
- Medical Devices
- OEM Medical Devices
- Product Introductions
- Protective Equipment
- Under Pads
- What are custom-made medical devices
- What are diagnostic products
- What are hospital dressing products
- What are medical tube catheters
- What are some common protective equipment
- What are the appropriate applications for hospital dressing products
- What are the appropriate uses for protective equipment
- What is a gynecological examination
- What is a medical consumable
- What is an anesthesia kit
- What is an OEM in medical devices
- what is an wound skin care
- what is can disposable ultrasonic diagnostic
- What is good manufacturing medical devices
- What is hospital-grade protective equipment
- what is medical equipments hospital furniture
- What is medical sterilization wrapping
- What is rehabilitation equipment device
- What medical consumables do you supply
- Where can I find laboratory consumables wholesale
- where can I find medical protection device
- where to buy hypodermic accessories
- where to buy medical apparel
- where to buy medical consumable accessories
- where to find OEM medical device supplier
- where to find rehabilitation equipment supplier