How to Choose Catheter Manufacturer: The 2026 Guide
Would you ever hire a pilot who had only practiced on a flight simulator, or a surgeon who’d only read about an operation in a book? Probably not. In the medical device industry, the stakes are just as high. A $0.50 difference in the unit price of a tube can easily lead to a $500,000 loss in clinical delays if your vendor fails a quality audit. If you’re currently trying to figure out how to choose catheter manufacturer, you aren’t just looking for a factory; you’re looking for an insurance policy against failure.
The reality is that catheter manufacturing is a complex marriage of polymer science and rigorous regulatory documentation. You need a partner who understands that "close enough" is a dangerous phrase in a cleanroom. Whether you're a startup or a global OEM, here is the technical roadmap to finding a partner that actually delivers.
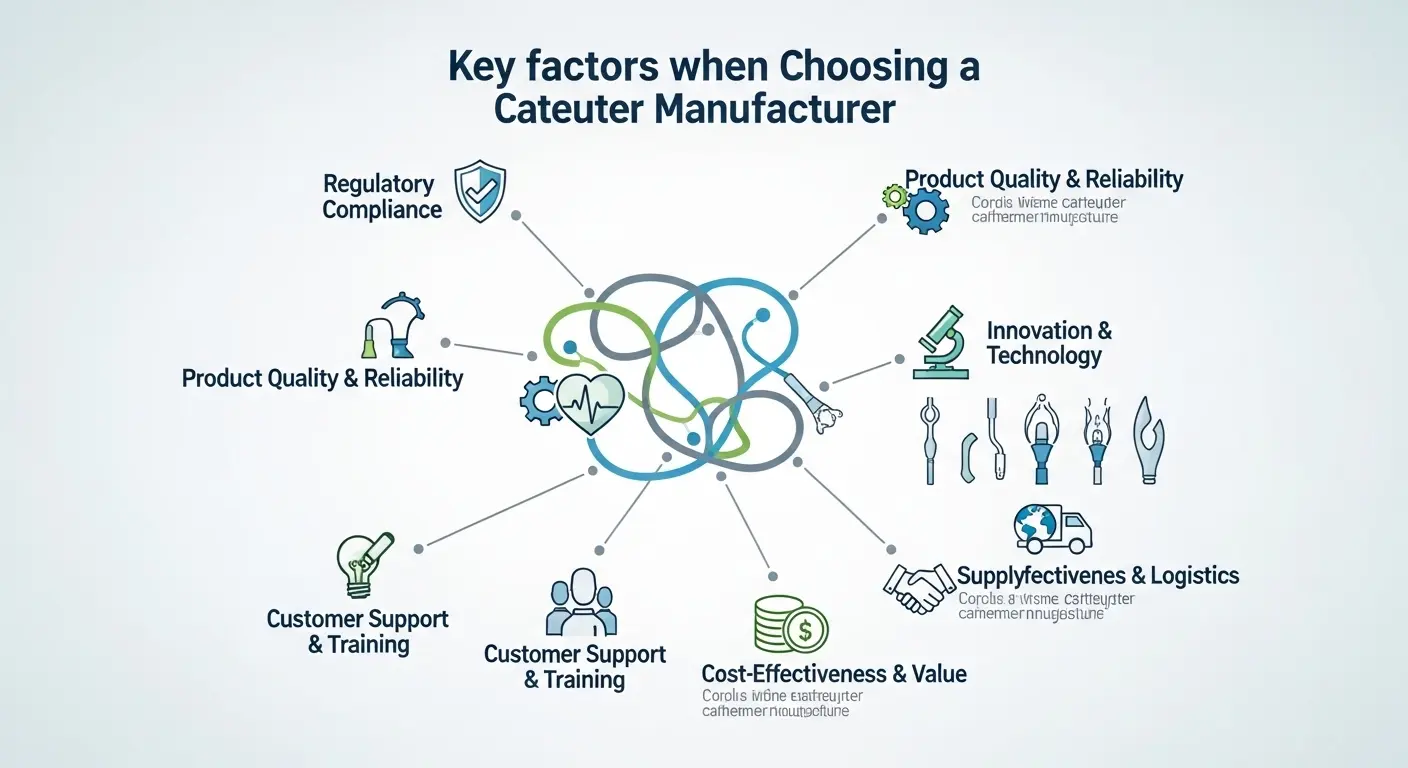
What are the 3 non-negotiables when choosing a catheter manufacturer?
When you evaluate potential partners, focus on three critical areas: ISO 13485 certification, vertical integration of extrusion and assembly, and proven Design for Manufacturability (DFM) expertise. A manufacturer must have a robust Quality Management System (QMS) to ensure every part is traceable back to the raw resin lot.
Additionally, vertical integration is vital. Working with a partner who handles both the extrusion and the secondary tipping or braiding reduces your supply chain risk. Finally, DFM expertise ensures that your design isn't just a "pretty drawing" but is actually optimized for high-yield, cost-effective mass production.
The Technical Audit: 5 Metrics That Actually Matter
Don't let a glossy brochure fool you. To truly understand how to choose catheter manufacturer, you have to look at the raw data. Here are the five engineering metrics that separate the leaders from the laggards.
1. Concentricity and Ovality

If the hole in the middle of your tube isn't perfectly centered, you have a problem. This is called concentricity. Low concentricity creates "thin spots" that are prone to bursting under high pressure. Similarly, ovality (how circular the tube is) affects how well the catheter bonds to connectors.
2. Surface Friction Coefficient
How well does the catheter slide? In neurovascular or cardiovascular cases, lubricity is king. You’ll need to decide between a PTFE (Teflon) liner or a hydrophilic coating. Your manufacturer should be able to provide the "Gram-Force" data for their surface finishes.
3. Bond Integrity
A catheter is often made of several different materials fused together. Specifically, you need to check the bond strength between the stiff shaft and the soft, atraumatic tip. We use RF-welding and laser-bonding to ensure these joints never fail inside a patient.
4. Dimensional Stability (CpK)
Ask for their Process Capability Index (CpK). This number tells you if their machines can stay within your specified tolerances for an entire 10,000-meter run. Specifically, if their CpK is lower than 1.33, you’re looking at a high risk of "quality drift."
5. Material Library Access
A great manufacturer should have a deep bench of medical-grade polymers. From Pebax® and TPU to Nylon 12 and PEEK, they should know exactly how each material behaves during the cooling phase of extrusion.
For instance, the structural stability needed for a specialized malecot catheter is entirely different from a standard Foley. Your partner must understand these nuances.
The Regulatory Shield: Navigating Standards in 2026
In 2026, the regulatory landscape is more unforgiving than ever. With the full implementation of the EU's Medical Device Regulation (MDR) and the FDA’s tightening of 510(k) pathways, your manufacturer is your first line of defense.
You should ask to see their validation packages. A reputable OEM will provide IQ/OQ/PQ (Installation, Operational, and Performance Qualification) data as part of the project. This documentation is essentially the "passport" your device needs to enter the market. Without it, you’ll be stuck in a cycle of regulatory rejections.
This level of documentation mirrors the high standards found in manufacturing a high-quality medical hypodermic syringe, where even a microscopic particulate can halt an entire production line.
Scaling Without Stalling: Prototype to Mass Production
Most boutique shops can make ten "perfect" samples in a lab. However, can they make 10,000 units with the same level of precision? This is what we call the "Transition Trap."
When you’re figuring out how to choose catheter manufacturer, look for a partner that handles both R&D and high-volume production. This "vertically integrated" approach allows the same engineers who built your prototype to oversee the scale-up. Consequently, you avoid the "lost in translation" errors that happen when you move between different vendors.
At Greetmed, we apply this philosophy across our entire range. Whether we're developing a complex interventional delivery system or supplying high-volume hospital dressing products, our goal is always to move from "concept to clinical" as fast as possible without sacrificing safety.
FAQ: Common Questions About Manufacturer Selection
How do I verify a manufacturer's ISO 13485 certificate?
Always ask for the full certificate and check the "Scope" of the audit. Ensure it specifically covers "Design and Manufacturing" of medical devices. Furthermore, verify the certificate number with the issuing body (like TUV or BSI) to ensure it hasn't expired.
What is the difference between an OEM and a CM?
A Contract Manufacturer (CM) simply builds exactly what you tell them to. An Original Equipment Manufacturer (OEM) like Greetmed provides the engineering expertise to help you improve the design, select materials, and manage the regulatory filing.
How does "PRN" apply to catheter supply chains?
In clinical logistics, some items are stocked as "as needed." If you’ve ever wondered what PRN means in medical terms, it essentially allows for flexible supply chain management in hospitals. A good manufacturer helps you manage these "as needed" surges by keeping a robust buffer of raw materials.
What is the typical lead time for a new catheter prototype?
For most "Design-to-Print" projects, you should expect to see your first sterile prototypes in 4 to 8 weeks. If the project requires custom extrusion tooling or exotic polymers, it might stretch to 12 weeks.
How can I check the cleanliness of a factory from across the world?
Ask for their "Particulate Count" reports and photos of their gowning protocols. A true Class 100,000 (ISO 8) or Class 10,000 (ISO 7) cleanroom will have strict, documented airflow and filtration logs that they should be happy to share.
Is a cheaper price always a red flag?
Not necessarily, but you have to look at why it’s cheaper. Is it because they have a more efficient supply chain, or because they aren't performing 100% in-line inspection? Never sacrifice inspection protocols for a lower unit cost. Even a simple medical cotton swab can cause infection if the manufacturing environment isn't controlled.
Your Engineering Ally
Choosing a partner is a long-term commitment. Don't just settle for a vendor who sends you a quote; find a partner who asks you hard questions about your design. Precision isn't just about the machines on the floor; it’s about the mindset of the people running them.
At Greetmed, we take pride in being the bridge between complex engineering and global scalability. We know that behind every tube we extrude is a patient who depends on our accuracy. Let's make sure your device is ready for the challenge.
Submit Your Request
Recent Posts
Tags
- Adult Diapers
- Are custom medical devices safe
- Baby Diapers
- Can respiratory anesthesia be used
- Digital Healthcare
- Do you offer customized consumables
- European Market
- How do you take care of a skin wound
- Industry Trends
- Lady Sanitary Napkins
- Medical Devices
- OEM Medical Devices
- Product Introductions
- Protective Equipment
- Under Pads
- What are custom-made medical devices
- What are diagnostic products
- What are hospital dressing products
- What are medical tube catheters
- What are some common protective equipment
- What are the appropriate applications for hospital dressing products
- What are the appropriate uses for protective equipment
- What is a gynecological examination
- What is a medical consumable
- What is an anesthesia kit
- What is an OEM in medical devices
- what is an wound skin care
- what is can disposable ultrasonic diagnostic
- What is good manufacturing medical devices
- What is hospital-grade protective equipment
- what is medical equipments hospital furniture
- What is medical sterilization wrapping
- What is rehabilitation equipment device
- What medical consumables do you supply
- Where can I find laboratory consumables wholesale
- where can I find medical protection device
- where to buy hypodermic accessories
- where to buy medical apparel
- where to buy medical consumable accessories
- where to find OEM medical device supplier
- where to find rehabilitation equipment supplier