PEBAX Tubing for Catheters: Complete Material Guide for OEM Medical Device Engineers
If you're designing or sourcing a catheter — whether it's a guide catheter, diagnostic catheter, balloon catheter, or neurovascular device — there is one material engineers consistently reach for first: PEBAX tubing.
It's not hype. PEBAX (polyether block amide) has become the industry standard for catheter shafts and tips because it solves a problem that most other polymers can't: delivering a wide, tunable stiffness range in a single biocompatible, kink-resistant material. From ultra-soft 25D distal tips to rigid 74D proximal shafts, PEBAX does what no single alternative can replicate on its own.
This guide covers everything you need to make an informed material decision — grade selection, real-world catheter applications, direct comparisons with competing materials, sterilization and compliance data, and how to work with a China-based OEM manufacturer like Greetmed to bring your design into production.
What Is PEBAX Tubing?

PEBAX is the brand name for polyether block amide (PEBA), a thermoplastic elastomer originally developed and commercialized by Arkema. It belongs to a family of block copolymers where hard polyamide segments (nylon-like, for stiffness) alternate with soft polyether segments (for elasticity and flexibility). By adjusting the ratio of these two blocks, manufacturers can produce grades that range from rubber-soft to near-rigid — all within the same material family.
For catheter manufacturing, this tunability is invaluable. A single catheter shaft can use two or three different PEBAX grades in a dual-durometer or tri-durometer extrusion — soft at the distal tip to prevent vessel trauma, progressively stiffer toward the hub for pushability and torque control. No other polymer class achieves this transition as cleanly or predictably.
Key intrinsic properties of PEBAX for medical applications:
- Shore D hardness range from 25D (soft elastomer) to 74D (semi-rigid)
- Excellent kink resistance at very thin wall thicknesses
- High torque transmission relative to wall thickness
- Softens slightly at body temperature (37°C), reducing tissue trauma
- Superior hydrolytic stability — does not absorb moisture and weaken over time
- Bonds well to adhesives, coatings (hydrophilic, PTFE), and overmolded components
- Biocompatible per ISO 10993 — suitable for blood-contacting applications
PEBAX Grade Selection Guide for Catheter Engineers
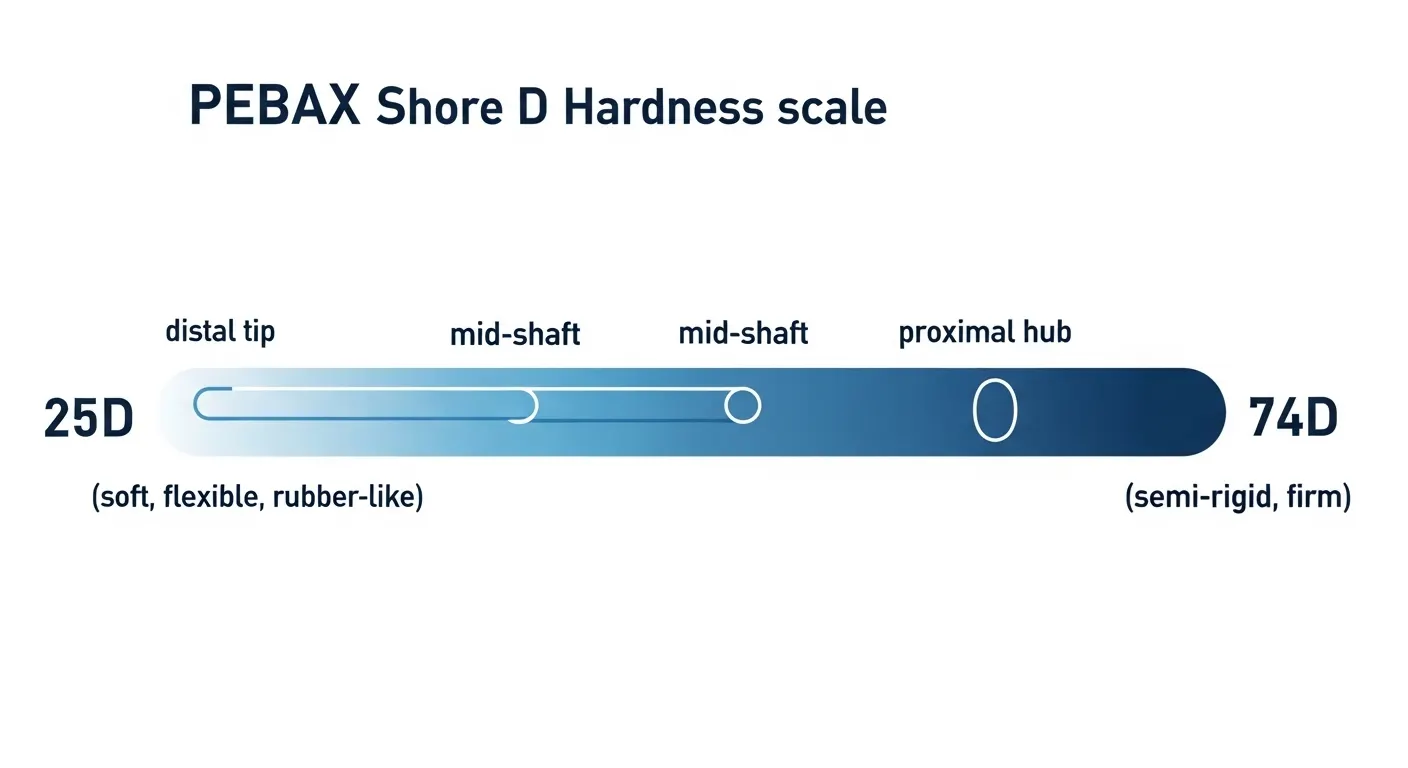
This is where most published material stops short. Knowing PEBAX is good for catheters is not enough — you need to match the right grade to the right segment of your device.
Here is a practical grade selection reference for catheter applications:
| PEBAX Grade | Shore D Hardness | Recommended Catheter Segment / Application |
|---|---|---|
| Pebax 2533 | 25D | Ultra-soft atraumatic distal tips, floppy guide wire lumen tips |
| Pebax 3533 | 35D | Flexible distal catheter body, pediatric catheter shafts |
| Pebax 4033 / 4533 | 40–45D | Mid-shaft flexible transition zones, balloon catheter bodies |
| Pebax 5533 | 55D | Intermediate transition zones in multi-durometer constructions |
| Pebax 6333 | 63D | Guide catheter distal-mid shafts, EP catheter shafts |
| Pebax 7033 / 7233 | 70–72D | Diagnostic catheter outer jackets, intermediate guide catheter sections |
| Pebax 7433 | 74D | Proximal stiff hub segments, pushability zones, outer jackets |
A practical design rule: The distal third of a catheter typically uses 25D–40D grades; the middle third uses 55D–63D; the proximal third uses 70D–74D. Transitions between grades can be achieved via heat-bonding, adhesive bonding, or coextrusion depending on your manufacturing process and tolerance requirements.
Catheter Types Greetmed Supplies PEBAX Tubing For
At Greetmed, our Medical Tube & Catheter manufacturing program covers a broad range of catheter types, each with its own PEBAX specification requirements:
Guide Catheters
Guide catheters require high pushability and axial stiffness in the proximal shaft combined with a softer distal tip for atraumatic vessel engagement. Typical construction uses Pebax 7433 (proximal) transitioning to Pebax 3533 or 4533 (distal). Braided reinforcement layers are commonly incorporated for added torque response.
Diagnostic Catheters
Precision torque transmission is the primary requirement here. Diagnostic catheters need thin-walled, tightly dimensioned tubing across the full shaft — typically Pebax 7033 or 7233 for the outer jacket — with tight ID/OD tolerances to maintain catheter curve memory under rotation.
Balloon Catheters
Balloon catheters often use PEBAX in semi-compliant balloon construction (Pebax 4033–5533) as well as the shaft body. PEBAX's flexibility-to-strength ratio makes it ideal for thin-walled balloon membranes that need to expand predictably without rupture.
Electrophysiology (EP) Catheters
EP catheters demand controlled deflection and lateral flexibility. Pebax 5533 to 6333 grades are common for the catheter shaft, with pull-wire channels precisely positioned in the lumen layout. The material's consistency at body temperature is particularly important for repeatable deflection curves.
Neurovascular / Microcatheters
These represent the most demanding application. Neurovascular catheters navigate tight intracranial vessels, requiring extreme flexibility and the softest possible tip (Pebax 2533 or 3533) with near-zero kink risk. Wall thicknesses as thin as 0.003" are achievable with the right extrusion setup.
Urological Catheters
While urological catheters — including the Nelaton catheter and latex urethral catheter formats we supply — are often manufactured in PVC or silicone for cost reasons, PEBAX is specified in premium intermittent and hydrophilic-coated urological catheter designs where kink-free flexibility and smooth surface adhesion are priorities. You can explore our broader urology and ostomy product line for more context.
PEBAX vs. Other Catheter Tubing Materials
Medical device engineers regularly evaluate PEBAX against Nylon (PA12), Polyurethane (PU), and fluoropolymers like PTFE/FEP. Here is an honest side-by-side:
| Property | PEBAX | Nylon (PA12) | Polyurethane (PU) | PTFE / FEP |
|---|---|---|---|---|
| Durometer range | 25D–74D (continuous) | Limited (typically 60D+) | Moderate range | Rigid |
| Kink resistance | ✅ Excellent | Good | Fair | Poor |
| Biocompatibility | ✅ ISO 10993 | ✅ | ✅ | ✅ |
| Adhesion / bondability | ✅ Good | Moderate | Good | ❌ Difficult |
| Torque transmission | ✅ High | High | Moderate | Low |
| Body temp. softening | ✅ Yes (reduces trauma) | Minimal | Slight | None |
| Hydrophilic coating adhesion | ✅ Excellent | Good | Good | ❌ Poor |
| Sterilization compat. | ✅ EtO, Gamma, E-Beam | ✅ | Gamma only | ✅ |
| Multi-durometer extrusion | ✅ Yes | Limited | Limited | ❌ No |
| Cost (relative) | Mid | Low | Mid | High |
| OEM customization | ✅ Very high | Moderate | Moderate | Low |
Where PEBAX wins decisively: Flexibility range, multi-durometer design, hydrophilic coating adhesion, and kink resistance. Where alternatives may be preferred: Nylon PA12 for pure cost reduction in simple single-lumen designs; PTFE liner layers for lubricity inside lumen walls (PEBAX/PTFE coextrusion is a well-established design approach).
Sterilization Compatibility and Biocompatibility
For medical device procurement teams, material certification is non-negotiable — and it's a topic most supplier pages ignore entirely.
Sterilization methods compatible with PEBAX tubing:
- Ethylene Oxide (EtO): ✅ Fully compatible — the most common sterilization method for PEBAX catheter assemblies
- Gamma Irradiation: ✅ Compatible at standard medical doses (25–35 kGy); minimal effect on mechanical properties
- E-Beam: ✅ Compatible at controlled doses
- Steam / Autoclave: ❌ Not recommended — PEBAX's polyether segments are susceptible to hydrolytic degradation under prolonged high-temperature steam exposure
- Dry Heat: ❌ Not recommended
Biocompatibility documentation:
PEBAX grades used in medical devices are evaluated per ISO 10993 (Biological Evaluation of Medical Devices). Standard tests relevant to catheter applications include:
- ISO 10993-5: Cytotoxicity
- ISO 10993-10: Sensitization and irritation
- ISO 10993-4: Blood compatibility (for intravascular devices)
At Greetmed, our manufacturing is certified to ISO 13485, and our products carry CE marking and FDA clearances relevant to their device classifications. You can review our certification credentials on our About Us page. For specific device-level biocompatibility documentation, our engineering team can provide testing reports as part of the OEM qualification process.
The OEM Custom PEBAX Tubing Process at Greetmed
If you're an OEM engineer or procurement manager evaluating a new catheter tubing supplier, here is exactly what the engagement process looks like with Greetmed:
Step 1 — Submit Your Specifications
Provide your target dimensions (inner diameter, outer diameter, wall thickness), Shore D hardness, lumen count and configuration (single, dual, multi), preferred length, and any overmolding, braiding, or coextrusion requirements. If you don't have final specs yet, our engineering team can help you develop them.
Step 2 — Engineering Review and Material Recommendation
Our R&D team reviews your specs against our material library and manufacturing capabilities. If a single PEBAX grade satisfies your requirements, we confirm it. If your design requires variable stiffness or a coextruded construction, we propose the grade sequence and joining method.
Step 3 — Prototype and Sample Production
We produce physical samples for dimensional verification, flexibility assessment, and initial testing. Samples are shipped for your in-house evaluation. Most standard prototype runs are completed and shipped within 3–4 weeks.
Step 4 — Compliance and Documentation Package
We prepare or provide access to: material certificates of conformance (CoC), ISO 10993 test reports, dimensional inspection reports, and process validation documentation as required by your regulatory pathway (MDR, FDA 510(k), etc.).
Step 5 — Production Scaling and Delivery
Approved designs move to production. We ship from Shanghai and Ningbo ports to global destinations. OEM labeling and private-label packaging are available. Payment via T/T or L/C. Inquiries and supply questions are answered within 8 hours.
For a broader overview of how we handle distribution and supply chain management, see our Catheters and Tubing Distributor page.
Why Source PEBAX Catheter Tubing from a China OEM Manufacturer?

This is the question most Western engineers ask — and it deserves a direct answer.
US- and Europe-based PEBAX extruders are excellent, but they carry structural cost disadvantages: higher labor rates, higher overhead, and longer lead times for custom work due to production backlogs. For OEM companies running R&D programs, scaling from prototype to production, or managing cost-sensitive product lines, China-based GMP manufacturing closes that gap significantly without compromising compliance.
Here is what Greetmed specifically brings to the table:
- Established since 2003 — over 20 years of medical device manufacturing experience
- GMP-certified workshop across three manufacturing facilities in Hubei, Anhui, and Zhejiang provinces, covering more than 10,000 m² of production space
- ISO 13485 certified, with CE and FDA clearances across product categories
- MDR certifications — Greetmed is recognized as a Chinese industry leader in MDR-compliant medical consumables
- 200+ product categories — the same supplier that handles your PEBAX catheter tubing can also supply your catheter care accessories, wound care, respiratory, and other consumable needs
- OEM and private label production fully supported
- 8-hour inquiry response policy — no waiting days for a quote
For medical device companies managing multiple SKUs and international supply chains, working with a single certified OEM partner reduces audit burden, simplifies logistics, and accelerates time-to-market.
Frequently Asked Questions
Pebax 63D (Pebax 6333) is softer and more flexible — it's typically used for guide catheter distal and mid shafts where flexibility is needed without sacrificing torque response. Pebax 7233 is stiffer and provides better pushability, making it better suited for proximal shaft segments or outer jackets in diagnostic catheters where column strength matters more than flexibility.
Yes. Medical-grade PEBAX grades are evaluated per ISO 10993 and are widely used in blood-contacting intravascular devices including coronary guide catheters and neurovascular microcatheters. Device-level biocompatibility testing (cytotoxicity, sensitization, blood compatibility) is required regardless of material compliance, and we can support your testing program with material documentation.
Yes. PEBAX coextrusion is well-established in catheter manufacturing. Common constructions include PTFE/PEBAX dual-layer tubing (PTFE inner liner for lubricity, PEBAX outer for flexibility), and nylon/PEBAX combinations. Multi-lumen coextrusions are also achievable for EP catheters and complex endoscopic devices.
Ethylene Oxide (EtO) is the most widely used and fully compatible method. Gamma and e-beam irradiation are also compatible. Autoclaving is not recommended due to the risk of hydrolytic degradation in the polyether segments.
MOQ depends on the complexity of the design, lumen configuration, and dimensional specifications. Contact our team directly for a quotation — we accommodate both R&D-scale prototype runs and high-volume production orders.
Nylon 12 (PA12) is stiffer and less flexible than PEBAX at comparable wall thicknesses, and it offers a narrower hardness range. For single-zone guide catheter shafts where stiffness and cost are the primary drivers, Nylon 12 may be sufficient. For multi-zone variable-stiffness designs or when hydrophilic coating adhesion is required, PEBAX is consistently the better choice.
Yes. Hydrophilic coating adhesion is one of PEBAX's key advantages over PTFE and other low-surface-energy polymers. Our engineering team can discuss coating integration as part of the OEM development program.
Ready to Source PEBAX Tubing for Your Catheter Project?
Whether you're finalizing a catheter design, evaluating a new supplier, or scaling an existing product line, Greetmed has the manufacturing infrastructure, certification credentials, and technical expertise to support your program.
Explore our full Medical Tube and Catheter product range or contact us directly to discuss your specific PEBAX tubing requirements. Our team responds within 8 hours.
Submit Your Request
Recent Posts
Tags
- Adult Diapers
- Are custom medical devices safe
- Baby Diapers
- Can respiratory anesthesia be used
- Digital Healthcare
- Do you offer customized consumables
- European Market
- How do you take care of a skin wound
- Industry Trends
- Lady Sanitary Napkins
- Medical Devices
- OEM Medical Devices
- Product Introductions
- Protective Equipment
- Under Pads
- What are custom-made medical devices
- What are diagnostic products
- What are hospital dressing products
- What are medical tube catheters
- What are some common protective equipment
- What are the appropriate applications for hospital dressing products
- What are the appropriate uses for protective equipment
- What is a gynecological examination
- What is a medical consumable
- What is an anesthesia kit
- What is an OEM in medical devices
- what is an wound skin care
- what is can disposable ultrasonic diagnostic
- What is good manufacturing medical devices
- What is hospital-grade protective equipment
- what is medical equipments hospital furniture
- What is medical sterilization wrapping
- What is rehabilitation equipment device
- What medical consumables do you supply
- Where can I find laboratory consumables wholesale
- where can I find medical protection device
- where to buy hypodermic accessories
- where to buy medical apparel
- where to buy medical consumable accessories
- where to find OEM medical device supplier
- where to find rehabilitation equipment supplier