PFA vs RFA in Medical Device Quality Management: Key Differences and Best Practices
In highly regulated industries like medical device manufacturing, quality management frameworks are essential to ensure product safety, reliability, and compliance. Two commonly used quality investigation methods — Process Failure Analysis (PFA) and Root Failure Analysis (RFA) — help manufacturers identify problems, address systemic issues, and prevent recurrence.
This article explains the differences between PFA and RFA, how each is applied in medical device quality systems, and practical strategies for using both within structured frameworks such as ISO 13485, FDA QSR (21 CFR Part 820), and CAPA processes.
Why Failure Analysis Matters in Medical Device Manufacturing
Medical devices often serve life-critical or life-supporting functions. Failures — whether during production, testing, or clinical use — can have serious consequences for patients, end users, and brands. As a result:
-
Regulatory bodies expect robust quality systems that proactively identify and manage risks
-
Quality investigations are integral to CAPA (Corrective and Preventive Action) systems
-
Analysis outcomes must be documented and integrated into process improvements
Two analytical approaches that help organizations understand failures and corrective actions are Process Failure Analysis (PFA) and Root Failure Analysis (RFA).
What Is Process Failure Analysis (PFA)?
Process Failure Analysis (PFA) focuses on identifying process-related weaknesses or deviations in the manufacturing or quality system that contribute to failures. Rather than immediately examining the product itself, PFA examines the processes that produced the product.
Core Focus of PFA
PFA investigates how and why a manufacturing process, inspection routine, or quality checkpoint did not perform as intended. It aims to improve processes, reduce variability, and prevent systematic errors.
PFA typically includes investigation of:
-
Stage-gates in production workflows
-
Operator instructions and training gaps
-
Machine calibration and maintenance logs
-
Environmental conditions (cleanroom, humidity, temperature)
-
Test and inspection procedures
When to Use PFA
PFA is most useful when:
-
Failures correlate with specific process steps
-
Multiple units fail due to a common process issue
-
Process deviations are suspected (e.g., parameter drift, human error)
Example: If a batch of sterile packaging fails integrity tests, PFA might examine:
-
Sealer temperature stability
-
Operator handling procedures
-
Calibration records of sealing machines
-
Environmental monitoring during packaging
By focusing on the process, PFA helps teams reduce variability and strengthen production controls.
What Is Root Failure Analysis (RFA)?
Root Failure Analysis (RFA) digs deeper to identify the fundamental cause of a failure event. RFA goes beyond surface symptoms, asking why a failure occurred and systematically tracing that failure back to its underlying reason.
Core Focus of RFA
RFA is about uncovering the “true root cause” among multiple possible contributing factors. It often applies structured techniques such as:
-
5 Whys analysis
-
Fishbone (Ishikawa) diagrams
-
Fault tree analysis
-
Failure mode and effects analysis (FMEA)
RFA focuses on:
-
Design flaws
-
Material defects
-
Human factors
-
Systemic gaps in documentation or training
-
Inadequate risk controls
When to Use RFA
RFA is appropriate when:
-
Failures are complex or persistent
-
PFA did not reveal a definitive cause
-
Failures occur across multiple process steps or products
Example: If a diagnostic test consistently yields inaccurate results, RFA might explore:
-
Sensor calibration and firmware interpretation
-
Component tolerances and drift over time
-
Design assumptions under real-world usage
-
Validation gaps in environmental conditions
By identifying the true causal factor, RFA supports long-term corrective and preventive actions that eliminate recurrence.
Key Differences Between PFA and RFA
Understanding how PFA and RFA differ in objectives, methods, and outcomes is essential for applying them effectively.
| Aspect | Process Failure Analysis (PFA) | Root Failure Analysis (RFA) |
|---|---|---|
| Primary Focus | Process deviations and controls | Fundamental cause of failure |
| Scope | Production and quality processes | Product design, materials, systems |
| Typical Size of Issue | Isolated or process-linked | Persistent, recurring, complex |
| Approach | Process optimization | Causal discovery |
| Tools | SOP reviews, calibration checks | 5 Whys, Fishbone, FMEA |
| Outcome | Better process controls | Elimination of root causes |
In essence, PFA answers “What part of the process failed?”, while RFA answers “Why did that failure happen in the first place?”
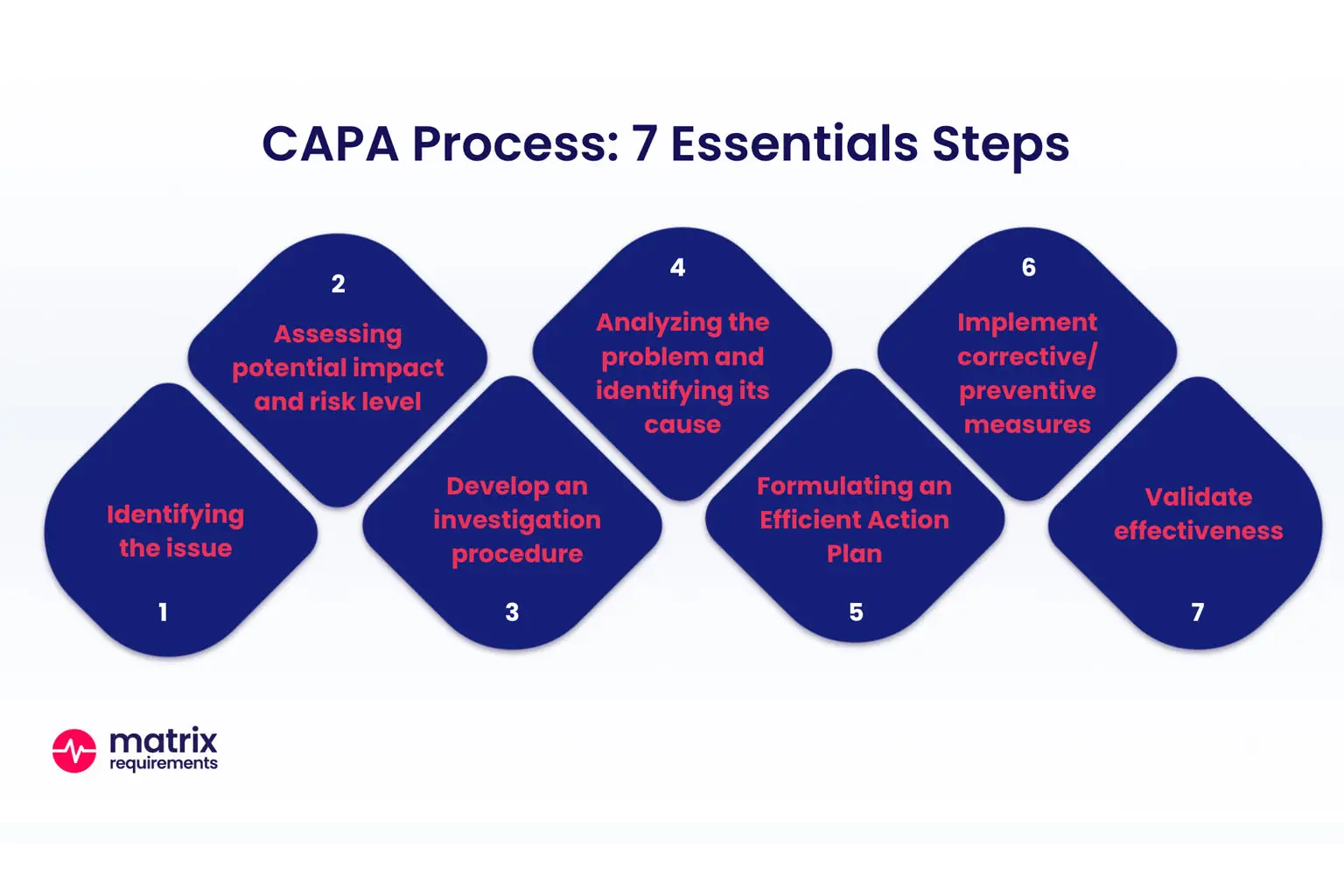
Integrating PFA and RFA Into CAPA Systems
Both PFA and RFA play vital roles in comprehensive CAPA (Corrective and Preventive Action) processes under quality systems such as ISO 13485 and FDA QSR.
1. Trigger Event
A non-conformance, complaint, audit finding, or field issue triggers an investigation.
2. Initial Assessment
Quick assessment determines whether the issue is likely due to process deviation, design flaw, or both.
3. PFA First
Start with PFA to evaluate if a process inconsistency is the most immediate source.
4. RFA for Deep Cause
If PFA does not yield a complete explanation or if failures persist, conduct RFA using structured methodologies.
5. Action Planning
Develop corrective actions that address:
-
Process parameter controls (from PFA)
-
Design, material, or systemic controls (from RFA)
6. Verification of Effectiveness
Use post-implementation metrics to confirm that corrective actions are effective and sustainable.
Integrating both analyses within CAPA ensures that both operational faults and deeper systemic causes are identified and mitigated.
Tools and Techniques for Effective Analysis
Here are commonly used tools that support PFA and RFA workflows:
5 Whys Analysis
A simple yet powerful technique that asks “Why?” repeatedly (often five times) until the fundamental cause is identified.
Fishbone (Ishikawa) Diagrams
Visual categorization of possible causes across areas such as Methods, Machines, Materials, and Manpower.
Failure Mode and Effects Analysis (FMEA)
A proactive method to identify potential failure modes, assess their severity, and prioritize controls before failures occur.
Fault Tree Analysis
A top-down logical approach that uses Boolean logic to trace failure paths.
These methods help quality teams structure thinking and avoid superficial conclusions.
Documentation and Traceability Requirements
Regulatory expectations — by agencies such as the FDA, EU Notified Bodies, and NMPA — require documented evidence of analysis. Appropriate documentation includes:
-
Investigation plans
-
Data collection and test reports
-
Analysis outputs (e.g., fishbone charts, 5 Whys trees)
-
Action plans
-
Verification of effectiveness reports
A well-structured report supports audits, inspections, and supplier communication.
Common Pitfalls and How to Avoid Them
Overlooking Systemic Causes
Teams sometimes stop after finding an apparent cause without exploring deeper systemic issues.
Solution: Use RFA tools that encourage deeper inquiry and challenge assumptions.
Inadequate Data Collection
Insufficient data can lead to incorrect conclusions.
Solution: Gather detailed quantitative and qualitative data from production runs, test results, and environmental conditions.
Insufficient Cross-Functional Involvement
Quality, engineering, manufacturing, and regulatory teams must collaborate for meaningful analysis.
Solution: Use cross-disciplinary teams to bring diverse perspectives to investigations.
Practical Example — Cap Analysis in Medical Device Manufacturing
Imagine a scenario where packages of sterile components show compromised seals during inspection:
-
PFA Approach: Evaluate sterilization process parameters, sealing machine calibration, operator training records, and environmental controls during packaging. If an out-of-tolerance parameter is found (e.g., inconsistent sealing temperature), process controls can be adjusted.
-
RFA Approach: If sealing parameters are within control limits but the issue persists, RFA explores deeper causes such as material compatibility with sterilization methods, packaging design flaws, incorrect tolerance specifications, or sensor calibration drift.
Combined, both analyses lead to corrective action that addresses both process and systemic causes.
Conclusion — Building a Resilient Quality Culture
In regulated medical device manufacturing, failures are not just problems to fix — they are opportunities to improve systems and strengthen compliance. Process Failure Analysis (PFA) and Root Failure Analysis (RFA) each play distinct but complementary roles in quality investigations.
PFA helps refine and control production processes, while RFA identifies deeper systemic causes that, if left unaddressed, can lead to persistent or recurring issues. By integrating both approaches into a structured CAPA framework, manufacturers ensure higher quality, greater product reliability, and stronger regulatory readiness.
Deploying structured analysis tools, engaging cross-functional teams, and maintaining rigorous documentation will not only satisfy compliance authorities but also foster a proactive culture that anticipates risks and drives continuous improvement.
Submit Your Request
Recent Posts
Tags
- Adult Diapers
- Are custom medical devices safe
- Baby Diapers
- Can respiratory anesthesia be used
- Digital Healthcare
- Do you offer customized consumables
- European Market
- How do you take care of a skin wound
- Industry Trends
- Lady Sanitary Napkins
- Medical Devices
- OEM Medical Devices
- Product Introductions
- Protective Equipment
- Under Pads
- What are custom-made medical devices
- What are diagnostic products
- What are hospital dressing products
- What are medical tube catheters
- What are some common protective equipment
- What are the appropriate applications for hospital dressing products
- What are the appropriate uses for protective equipment
- What is a gynecological examination
- What is a medical consumable
- What is an anesthesia kit
- What is an OEM in medical devices
- what is an wound skin care
- what is can disposable ultrasonic diagnostic
- What is good manufacturing medical devices
- What is hospital-grade protective equipment
- what is medical equipments hospital furniture
- What is medical sterilization wrapping
- What is rehabilitation equipment device
- What medical consumables do you supply
- Where can I find laboratory consumables wholesale
- where can I find medical protection device
- where to buy hypodermic accessories
- where to buy medical apparel
- where to buy medical consumable accessories
- where to find OEM medical device supplier
- where to find rehabilitation equipment supplier