From Concept to Market: Medical Device Product Development Process Explained
Bringing a medical device from a concept to a commercially released product is a complex, multi-phase journey involving clinical insights, engineering rigor, regulatory compliance, and manufacturing readiness. Unlike consumer electronics or general industrial products, medical devices must demonstrate both safety and effectiveness in real-world clinical environments — making the development process structured, evidence-based, and highly regulated.
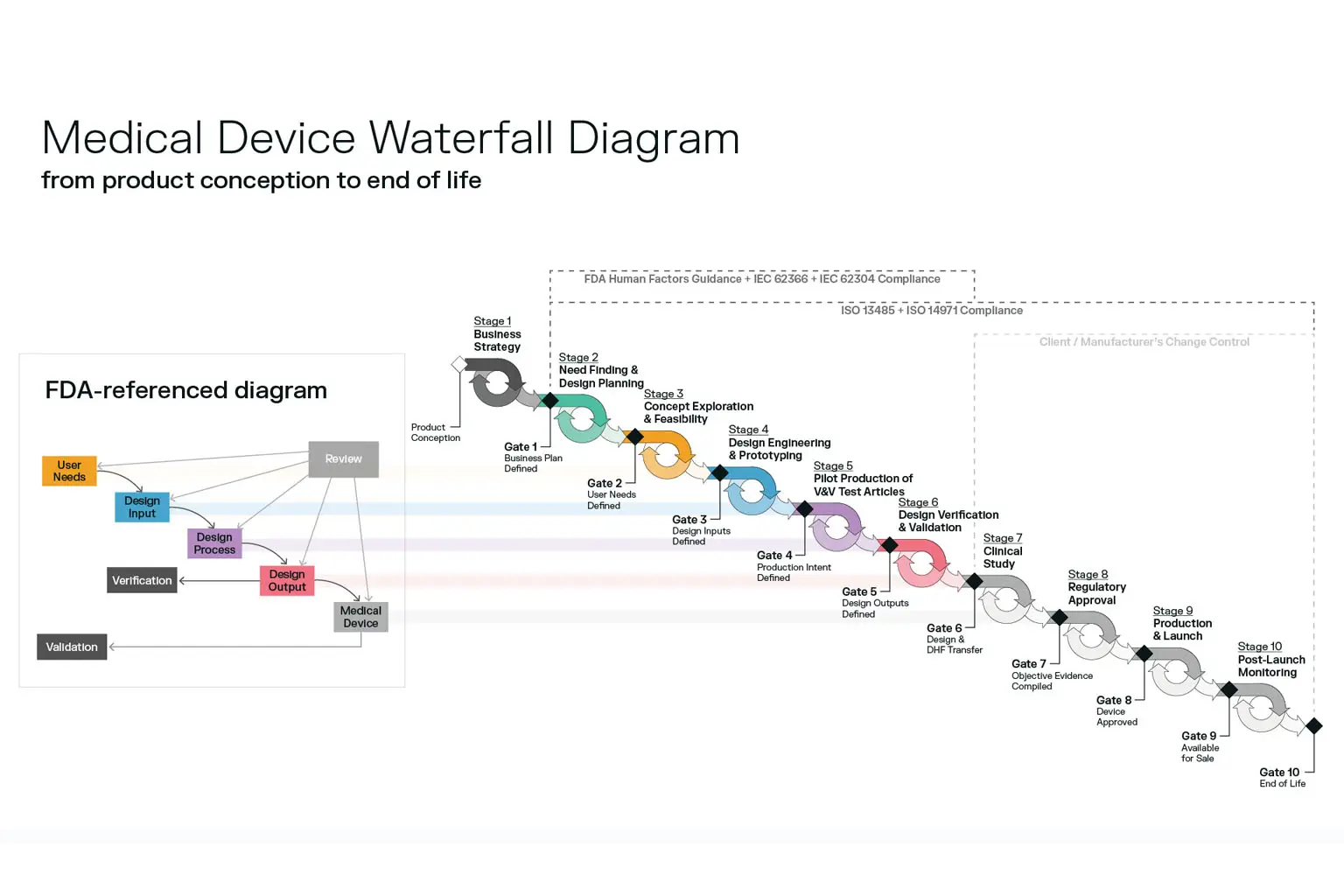
This article outlines the essential steps in the medical device product development process, from early idea generation to market launch and post-market support, helping OEMs, innovators, and quality teams understand key requirements and best practices for success.
Overview: Why a Structured Development Process Matters
Medical device development operates within an ecosystem of regulatory frameworks, quality standards, clinical expectations, and market needs. Whether you're developing an in-vitro diagnostic, a surgical instrument, or an implantable orthopedic device, a structured approach:
-
Reduces risk by identifying issues early
-
Supports regulatory approval with robust evidence
-
Aligns engineering and clinical requirements
-
Ensures manufacturability and scalability
-
Enhances user safety and market adoption
Frameworks such as ISO 13485 Design Controls, FDA 21 CFR Part 820, EU MDR, and other global regulatory guidelines embed development activities with checkpoints that guide decisions throughout the product lifecycle.
1. Ideation and Clinical Needs Analysis
The first and arguably most critical stage in any medical product development process is understanding the clinical problem you aim to solve. This phase includes:
-
Identifying unmet clinical needs
-
Interviewing clinicians, caregivers, and end users
-
Reviewing existing solutions and market gaps
-
Assessing potential impact on patient outcomes
A formal Needs Assessment Document is typically the first design input artifact and serves as a foundation for all subsequent design decisions.
2. Concept Generation and Feasibility Assessment
Once clinical needs are identified, the next phase focuses on generating design concepts and conducting feasibility analysis. Activities in this phase include:
-
Brainstorming multiple design ideas
-
Sketching and conceptual modeling
-
Preliminary risk identification
-
Evaluating technology options (hardware, sensors, materials)
Feasibility studies help determine whether design concepts are realistic, manufacturable, and capable of achieving targeted performance benchmarks.
3. Design Inputs and Requirements Definition
Building on clinical needs and feasibility insights, this phase defines design inputs — measurable, verifiable requirements that the product must satisfy. These typically include:
-
Functional requirements (what the device must do)
-
Performance specifications (accuracy, reliability)
-
Regulatory constraints
-
User interface and ergonomics criteria
-
Environmental conditions (sterilization, temperature, storage)
Well-defined design inputs are essential for later verification and validation activities, ensuring traceability throughout development.
4. Risk Management Planning
Risk management is not optional in medical device development — it’s mandated by standards such as ISO 14971. In this stage, teams:
-
Identify potential hazards across the product lifecycle
-
Evaluate severity and likelihood
-
Implement risk control measures
-
Document residual risk and risk/benefit analyses
Risk planning procedures become essential artifacts during regulatory submissions, especially for medium- and high-risk products.
5. Detailed Design and Engineering Iterations
With design inputs and risk plans established, engineers create detailed designs. This includes:
-
Mechanical design and CAD modeling
-
Electrical and embedded software architecture
-
Material selection
-
Simulation and performance modeling
Rapid prototyping methods such as 3D printing and soft tooling help validate form, fit, and function. A series of iterations refine design choices, reducing risk before finalizing designs.
6. Verification and Validation (V&V)
Verification and validation are formal, documented activities that confirm whether:
-
The design was implemented correctly (Verification)
-
The final product meets user needs in real or simulated environments (Validation)
Verification uses engineering tests (bench tests, tolerance checks, automated inspections), while validation often includes clinical simulations, usability studies, and human factors testing.
These activities produce evidence that supports regulatory submissions and manufacturing readiness.
7. Regulatory Strategy and Submission Planning
Parallel with design activities, a regulatory strategy is developed. This includes:
-
Determining product classification (e.g., Class I, II, III in the U.S.)
-
Identifying required regulatory pathways (e.g., FDA 510(k), PMA, EU MDR conformity assessment)
-
Planning test protocols and documentation submissions
Early engagement with regulatory authorities, where appropriate, helps clarify expectations and reduces surprises during review cycles.
8. Design Transfer and Manufacturing Readiness
Transitioning from design to manufacturing involves Design Transfer, where:
-
Production processes are documented and controlled
-
Bills of materials (BOM) and assembly instructions are finalized
-
Manufacturing partners are qualified
-
Quality controls and inspection plans are established
Manufacturability assessments ensure that designs are optimized for reproducible, scalable production — reducing defects and supporting consistent quality.
9. Pre-Launch Activities: Pilot Runs and Market Preparation
Before full commercial release, pilot production runs are conducted to:
-
Validate production tooling
-
Confirm process controls
-
Identify assembly or test bottlenecks
-
Support inventory and supply chain planning
Concurrently, market preparation includes:
-
Developing marketing materials
-
Establishing distribution channels
-
Training sales and clinical support teams
10. Regulatory Submission and Approval
After gathering all necessary evidence (V&V reports, risk analyses, quality system records, labeling), regulatory submissions are compiled. Depending on region and device class, this may involve:
-
FDA 510(k) Notification or Premarket Approval (PMA)
-
EU MDR Technical File and CE Marking
-
China NMPA Registration
-
Other regional submissions (Health Canada, TGA, PMDA)
Regulatory review periods vary, but thorough documentation and early planning improve acceptance odds.
11. Commercial Launch and Market Entry
Once regulatory approval is granted, commercialization activities proceed:
-
Product launch announcements
-
Distribution rollout
-
Clinical adoption programs
-
Customer support infrastructure
Continued focus on quality ensures early complaints or post-market signals are monitored and addressed promptly.
12. Post-Market Surveillance and Lifecycle Management
Medical device development doesn’t end at launch. Standards and regulations require ongoing post-market surveillance, including:
-
Monitoring adverse events
-
Tracking real-world performance
-
Analyzing customer feedback
-
Updating labeling or design as needed
-
Implementing corrective and preventive actions (CAPA)
Lifecycle management also includes planning for future versions, feature enhancements, or geographic expansion.
Key Success Factors in Medical Device Development
• Cross-Functional Collaboration
Design, engineering, quality, regulatory, manufacturing, and commercial teams must align early and often.
• Documentation Discipline
Traceability matrices, design histories, risk logs, and test records support both compliance and operational excellence.
• Risk-Based Thinking
Embedding risk assessment and mitigation at every phase reduces late-stage surprises.
• Early Regulatory Engagement
Proactive strategy shortens review cycles and increases predictability.
• Market Validation
Understanding clinical workflows, user preferences, and competitive differentiators drives product relevance.
Conclusion
The journey from concept to market in medical device product development is rigorous, evidence-based, and multidisciplinary. By following structured phases — from clinical needs analysis and design to regulatory submission and post-market surveillance — manufacturers and OEM partners can build products that are not only safe and effective but also manufacturable, compliant, and commercially successful.
In an environment where patient outcomes and quality of care are paramount, a disciplined product development process supports innovation while safeguarding both users and brands.
Submit Your Request
Recent Posts
Tags
- Adult Diapers
- Are custom medical devices safe
- Baby Diapers
- Can respiratory anesthesia be used
- Digital Healthcare
- Do you offer customized consumables
- European Market
- How do you take care of a skin wound
- Industry Trends
- Lady Sanitary Napkins
- Medical Devices
- OEM Medical Devices
- Product Introductions
- Protective Equipment
- Under Pads
- What are custom-made medical devices
- What are diagnostic products
- What are hospital dressing products
- What are medical tube catheters
- What are some common protective equipment
- What are the appropriate applications for hospital dressing products
- What are the appropriate uses for protective equipment
- What is a gynecological examination
- What is a medical consumable
- What is an anesthesia kit
- What is an OEM in medical devices
- what is an wound skin care
- what is can disposable ultrasonic diagnostic
- What is good manufacturing medical devices
- What is hospital-grade protective equipment
- what is medical equipments hospital furniture
- What is medical sterilization wrapping
- What is rehabilitation equipment device
- What medical consumables do you supply
- Where can I find laboratory consumables wholesale
- where can I find medical protection device
- where to buy hypodermic accessories
- where to buy medical apparel
- where to buy medical consumable accessories
- where to find OEM medical device supplier
- where to find rehabilitation equipment supplier